Auto logout in seconds.
Continue LogoutThe Simmons Comprehensive Cancer Center (SCCC) at the University of Texas Southwestern Medical Center (UT Southwestern) noticed that fragmented ordering processes and unstructured lab results were preventing metastatic prostate cancer patients from getting guideline-recommended germline and somatic testing. To better identify and act on those patients, UT Southwestern redesigned its prostate cancer workflow by integrating genomic results directly into Epic®, adding real-time and backstop alerts, and reviving its molecular tumor board. In doing so, UT Southwestern improved identification of metastatic patients for referral, expanded access to appropriate targeted therapies and clinical trials, and created a model for scaling precision oncology across the health system.
The challenge
NCCN* Clinical Practice Guidelines In Oncology (NCCN Guidelines®)1,2 recommend that metastatic prostate cancer patients undergo germline and somatic testing for homologous recombination repair gene mutations (HRRm). However, clinicians in the Genitourinary Cancer Clinic at the University of Texas Southwestern Medical Center (UT Southwestern) Simmons Comprehensive Cancer Center (SCCC) noticed that fewer patients than expected were tested.3
*National Comprehensive Cancer Network®
"We realized we were missing patients — not because the data wasn’t there, but because the workflow didn’t connect the dots."
This gap in testing is not unique to UT Southwestern: An email survey of about 500 American Urological Association members found that on average, only 29% of patients were tested for HRRm and other genetic mutations in line with guidelines.4 Moreover, national data show germline testing continues to lag behind somatic testing.5 A national shortage of genetic counselors — which ended in 2024 — compounded these challenges, creating genetic counseling bottlenecks.
UT Southwestern found two main barriers to testing. First, oncologists often had incomplete visibility into patients’ family history, prior lab results, and other eligibility criteria, because many labs returned results as PDFs or faxed documents that were not searchable in the electronic health record (EHR). This situation led to missed opportunities to test eligible patients.
Second, UT Southwestern’s testing workflow was fragmented. Because germline and somatic testing had separate ordering processes and oncologists received limited clinical decision support (CDS), some patients received only one test when both were indicated. On top of that, UT Southwestern SCCC’s molecular tumor board (MTB) lacked the infrastructure to analyze molecular data, curbing oncologists’ access to actionable insights and multidisciplinary guidance.
To close the testing gap, providers needed a clear view of each patient’s lab results and family history, decision support for testing referrals, and a robust infrastructure to better analyze molecular data and make diagnostic decisions. While this case study centers on UT Southwestern’s efforts to detect HRRm, the approaches used apply to all biomarkers.
The organization
The UT Southwestern Medical Center, located in Dallas, is an academic medical center dedicated to converting biomedical research into cutting-edge clinical treatment. The Harold C. Simmons Comprehensive Cancer Center — a National Cancer Institute-designated comprehensive cancer center — offers patients greater access to clinical trials and therapies not available at other facilities.
The approach
UT Southwestern changed their workflow at the system level to help address under-referral of patients with prostate cancer to guideline-recommended germline and somatic testing. First, they integrated genomic data into the EHR, so results were searchable in the system and visible in patients’ individual electronic records.
Next, UT Southwestern built a dual alert strategy to help oncologists identify eligible patients. First, an in-line OurPractice Advisory (OPA) flags eligible prostate cancer patients in real time for genomic testing referral. To improve capture of patients not identified by the OPA, UT Southwestern embedded a macro in the pathology report. The macro adds text to the report reminding providers that tissue samples meeting certain criteria are eligible to be referred for genomic testing.
Finally, UT Southwestern revived and revamped their multidisciplinary MTB, expanding oncologists’ access to timely analysis of molecular data and enhanced clinical decision-making.
The result
Searchable lab results reduced time to testing and treatment for patients with prostate cancer and administrative burden for providers. The new MTB gives oncologists support for molecular data analysis. Finally, integrating genomic data into the EHR laid the foundation to help improve clinical decision making, clinical trial matching, and data analytics in the future.
Patients received somatic and germline testing
How UT Southwestern improved their prostate cancer workflow
UT Southwestern used a multipronged approach to embed genomic biomarker testing and decision-making in their prostate cancer workflow. To close gaps in testing and ensure timely, guideline-based care, UT Southwestern combined collaborative leadership, EHR integration, automated CDS, and consistent multidisciplinary review.
The four elements
Prior to the workflow redesign, most genomic test results arrived at UT Southwestern as PDFs or faxed reports, which were scanned into their Epic® system as static images. Static images are difficult to search, which slowed provider access to results and often lead to missed referrals or duplicate testing. Adding to the burden, providers had to log into different lab systems to view and track results. To address these challenges, UT Southwestern partnered with labs that had completed a high volume of germline and somatic testing; received positive feedback from clinicians; and gained proven experience integrating EHRs and returning results as structured, discrete data.
UT Southwestern chose to leverage functionality available through the existing EHR so clinicians can order genomic tests and view results. Returning results automatically populate the pathology and oncology views in the pathology tab of the patient’s chart in real time, so clinicians can see results without digging through images or multiple systems.
UT Southwestern also worked with preferred labs to bulk-load historical results, creating a longitudinal dataset for each patient. Because somatic and germline results are now stored in the EHR as discrete fields, providers can search the patient population for specific genetic alterations, such as HRRm. That way, UT Southwestern has the potential to identify a greater number of patients eligible for precision therapies and genomically selected clinical trials.
To maintain an efficient workflow, UT Southwestern monitors ordering patterns and gathers clinician feedback at regular governance meetings or through direct outreach. Using those insights, the health system continuously refines the order and review process for genomic testing.
Assess yourself: Integrated ordering and results in the EHR
Clinicians order biomarker tests and receive results outside the EHR system. | Clinicians order tests through the EHR but receive unstructured results outside the EHR. | The organization integrates ordering and structured test results into EHR. |
If structured EHR integration isn’t feasible, consider starting with PDF upload tracking and flagging in EHR notes, and work with 1–2 lab partners who already support Fast Health Interoperability Resources (FHIR). | ||
EHR view: Integrated genomic testing results in patient charts
The EHR integration centralized results in an oncology snapshot, giving providers a single, structured view of patients’ genomic testing status and eligibility for next steps.
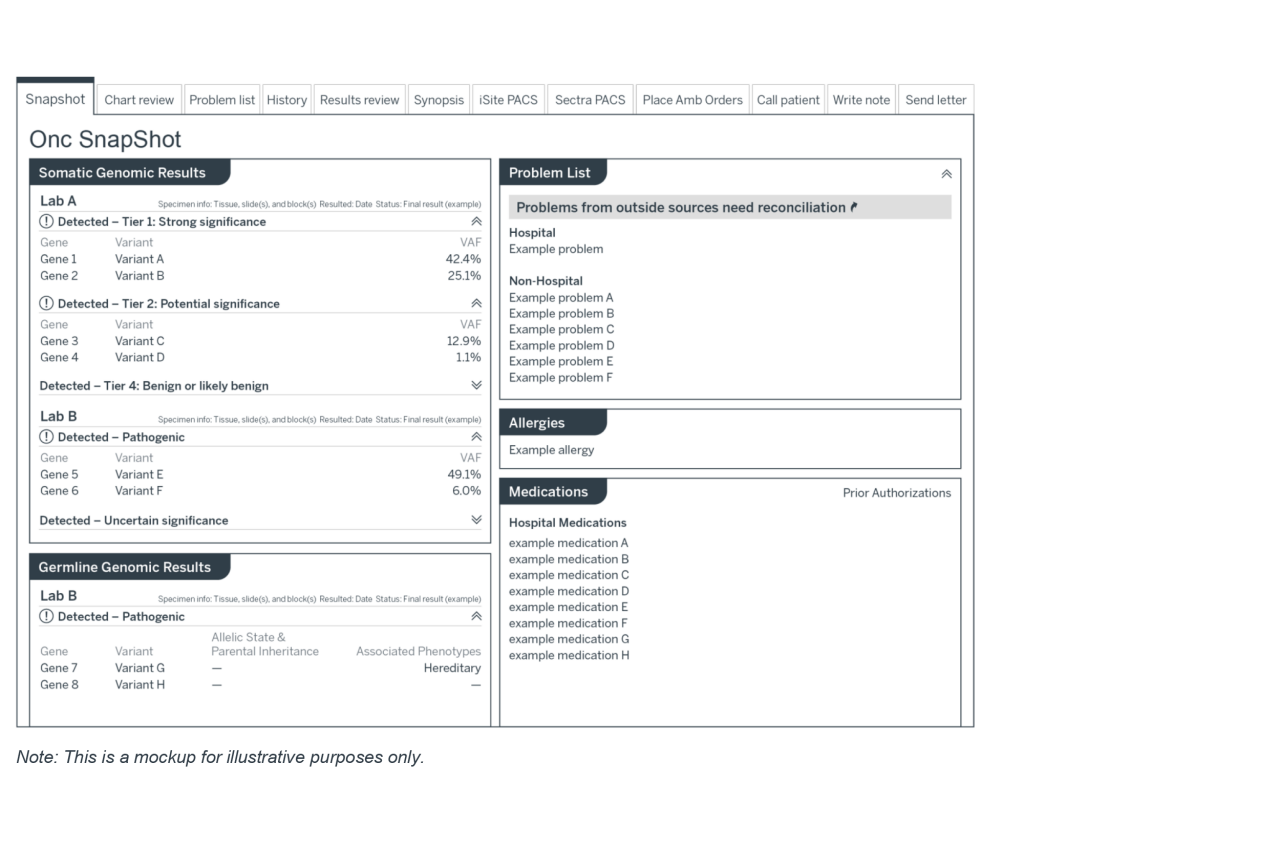
In 2022, about 50% of men with metastatic prostate cancer receive testing for HRRm. Of those, about 46% received somatic testing, 22% germline testing, and 32% both5 as guidelines recommend — highlighting the need to ensure that germline and somatic testing are ordered consistently. To close this gap, UT Southwestern built a dual alert strategy to improve capture of patients eligible for both. The system combines a real-time, in-line OPA with a macro embedded in the pathology notes. These two alerts allow clinicians to identify more patients flagged by NCCN Guidelines® criteria and those with high-grade tumors who might otherwise be missed.
EHR in-line OPA delivers real-time CDS for testing referrals
When a patient’s medical and family history is entered into the EHR, the system compares the patient’s information with National Comprehensive Cancer Network® (NCCN®)-based criteria for genomic testing. For example, a patient with a history of prostate cancer, a high Gleason score, and a maternal family member with breast cancer would meet three criteria (one criteria is enough to trigger testing).6 If a patient meets criteria but hasn’t been referred, an OPA pops up just before the provider signs the patient note and asks if they’d like to initiate a referral for testing. If the provider clicks “yes,” the referral is automatically placed.
The in-line OPA provides timely, minimally disruptive, and appropriate reminders and allows the team to monitor how often providers override or ignore alerts to improve messages and minimize alert fatigue. Since the system was implemented, there has been a 156% increase in referrals to genomic testing. Half of those referrals were a direct result of clicking the in-line OPA’s message.
In-line OPA criteria for patients with prostate cancer included:
|
Assess yourself: Integrated clinical decision support in EHRs
The organization does not have CDS related to genomic testing. | The EHR flags clinicians to test patients based on recommended guidelines. | The EHR prompts clinicians to wait for testing results and provides clinical recommendations based on the patient’s biomarkers. |
If your system doesn’t enable CDS, consider adding a free-text note template or macro in pathology reports to remind clinicians when a referral may be appropriate, using paper-based referral pathways, or implementing EHR smart phrases. | ||
Pathology macro captures patients missed by OPA criteria
Some patients have high-grade tumors but don’t trigger the in-line OPA criteria due to missing data such as family history. To capture these cases, in 2022 the team at UT Southwestern embedded a macro into the pathology note. When a Gleason score is 8 or greater, the macro inserts language into the note prompting providers to refer those patients for somatic or germline testing. While the macro isn’t as interactive as the in-line OPA, the text in the pathology note gives providers clinical decision support by reminding them when a referral is appropriate.
Using two entry points — a structured EHR history and pathology reporting — the alerts facilitate genomic testing, reduce the likelihood of missed referrals, and build confidence that a greater number of eligible patients are identified.
Pathology macro criteria for prostate cancer patients
|
Streamlining intake supports testing referrals and reduces bottlenecks
Before the workflow redesign, UT Southwestern SCCC’s molecular tumor board (MTB) met infrequently and had low attendance. Without an integrated infrastructure for analyzing biomarker results, providers had limited access to multidisciplinary expertise and struggled to act on findings. As part of the redesign, UT Southwestern restructured the MTB to include oncologists, molecular pathologists, surgeons, internal medicine physicians, genetic counselors, and trainees.
The MTB now meets biweekly to review the previous two weeks of complex solid tumor cases with genomic results and determine which are clinically actionable. Using Epic® reporting tools, the MTB filters results based on tumor type, gene, or therapeutic implication and highlights mutations and other molecular alterations with treatment potential, including rare or overlapping mutations that may not be highlighted in static reports, such as ATM or CDK12.
At MTB meetings, the board discusses patient cases, recommends treatment — including targeted therapies or clinical trials where appropriate — and documents guidance directly into the patient’s record. Oncologists from UT Southwestern satellite sites can either participate in the live discussion at the meeting or receive MTB recommendations via their EHR inbox, giving them broader access to expert input. Oncologists from satellite sites, fellows, and researchers may attend meetings to learn, present cases, and discuss research approaches.
Beyond individual case reviews, the MTB supports retrospective analyses, quality improvement projects, prevalence tracking, and equity-focused research initiatives, extending its impact across clinical care and research.
Assess yourself: Multidisciplinary forum to review genomic test results
The organization does not have a dedicated multidisciplinary group or process that discusses genomic test results. | The organization has a multidisciplinary forum or process to discuss day‑to‑day genomic testing cases, but often does not include all complex cases or clinicians from other system sites. | The organization has a multidisciplinary team (e.g., a molecular tumor board or genomics steering committee) or process that consistently reviews complex cases and discusses genomic testing cases and protocols with clinicians from satellite locations. |
If your system doesn’t have a molecular database, consider starting with a monthly MTB that is focused on 1–2 high-impact tumor types, and use PDF records and verbal case summaries. That’s enough to build clinician participation, partner with larger centers virtually, or allow regional clinicians to present cases. | ||
Results
The program has strengthened UT Southwestern SCCC’s ability to operationalize precision medicine by embedding somatic and germline workflows directly into clinical practice. By integrating genomic testing referral prompts and results into the EHR, UT Southwestern SCCC moved testing from an ad-hoc process to consistent, guideline concordant care for patients with prostate cancer.
Increase in referrals to somatic and germline testing since the in-line OPA was implemented. Half of those referrals were a direct result of clicking on the in-line OPA’s referral message
Patients received somatic and germline testing along with deep dive discussion and recommendations including precision oncology treatment approaches and clinical trial options over six months
Approximate relative increase in the share of tested prostate cancer patients with actionable HRR mutations identified following implementation of the pathology macro in 2022 (from ~6% to ~11%)
Evidence of impact: Workflow transformation
| Before | After | |
|---|---|---|
Referral and ordering |
|
|
Patient ID |
|
|
Workflow coordination |
|
|
Access to genetic counseling |
|
|
Data capture |
|
|
Clinical decision-making |
|
|
Patient impact |
|
|
1 Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Prostate Cancer V.5.2026 © National Comprehensive Cancer Network, Inc. 2026. All rights reserved. Accessed February 18, 2026. To view the most recent and complete version of the guideline, go online to NCCN.org.
2 NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in anyway.
3 Unless otherwise noted, all information in this case study came from Advisory Board interviews with faculty and staff from University of Texas Southwestern.
4 Practice Resource Library: Integrating Biomarker and Genetic Testing for HRR Mutations into Prostate Cancer Management. American Urological Association. Accessed September 5, 2025.
5 Barata, PC, et al. Genetic Testing in Men With Metastatic Castration-Resistant Prostate Cancer. JAMA Oncology. July 1, 2024.
6 Only one NCCN-based criterion must be met to trigger testing.
A note about this research
This example was developed to illustrate one healthcare organization's experience in developing and implementing an initiative to focus on improving patient care for patients with cancer. Pfizer did not provide direct financial support for the work completed. Participants from the health systems were compensated for their time in providing these insights.
All development and implementation, including health information technology (HIT) functionality, were solely chosen, implemented, and maintained by UT Southwestern. Pfizer does not endorse or recommend any specific functionality discussed within this resource. The health system is solely responsible for determining which EHR functionality to implement and for implementation of such functionality. All treatment decisions must be decided by the healthcare professional in consultation with the patient after review of the patient's full medical history.
This resource was not reviewed or endorsed by Epic®. All trademarks are the property of their respective owners. All information and data is provided by UT Southwestern and shared with permission. Pfizer does not guarantee any outcomes if health systems choose to implement a similar initiative.
At Pfizer, we’re in relentless pursuit of breakthroughs that change patients’ lives. We innovate every day to make the world a healthier place.
Our unique resources allow us to do more for people. Using our global presence and scale, we’re able to make a difference in local communities and the world around us.
Pfizer supports the global healthcare community’s independent quality improvement initiatives to improve patient outcomes in areas of unmet medical need that are aligned with Pfizer’s medical and/or scientific strategies. For more information, please visit www.pfizer.com.
This case study is sponsored by Pfizer, an Advisory Board member organization. Representatives of Pfizer helped select the topics and issues addressed. Advisory Board experts wrote the report, maintained final editorial approval, and conducted the underlying research independently and objectively. Advisory Board does not endorse any company, organization, product, or brand mentioned herein.
To learn more, view our editorial guidelines.

This case study is sponsored by Pfizer. Advisory Board experts conducted the underlying research independently and objectively.
Don't miss out on the latest Advisory Board insights
Create your free account to access 1 resource, including the latest research and webinars.
Want access without creating an account?
You have 1 free members-only resource remaining this month.
1 free members-only resources remaining
1 free members-only resources remaining
You've reached your limit of free insights
Become a member to access all of Advisory Board's resources, events, and experts
Never miss out on the latest innovative health care content tailored to you.
Benefits include:
You've reached your limit of free insights
Become a member to access all of Advisory Board's resources, events, and experts
Never miss out on the latest innovative health care content tailored to you.
Benefits include:
This content is available through your Curated Research partnership with Advisory Board. Click on ‘view this resource’ to read the full piece
Email ask@advisory.com to learn more
Click on ‘Become a Member’ to learn about the benefits of a Full-Access partnership with Advisory Board
Never miss out on the latest innovative health care content tailored to you.
Benefits Include:
This is for members only. Learn more.
Click on ‘Become a Member’ to learn about the benefits of a Full-Access partnership with Advisory Board
Never miss out on the latest innovative health care content tailored to you.

