Auto logout in seconds.
Continue Logout- Biomarker testing (also known as mutation, genomic, or molecular testing) uses laboratory tests to help the health care team gather as much information as possible about a patient’s cancer and uncover whether the patient has an actionable driver mutation.
- Biomarker testing can help providers make more informed treatment decisions based on a patient’s genomic makeup. Additionally, biomarker testing can align with a health system’s broader strategic goals (e.g., precision medicine).
A biomarker is a characteristic that can be scientifically measured or evaluated as an indicator of normal biologic processes, disease, or response to therapeutic intervention. The major types of biomarkers include molecular, histologic, radiographic, and physiologic characteristics. Examples of biomarkers include blood pressure, blood glucose levels, and imaging results. As the field of precision medicine grows, the focus on genetic biomarkers has grown as well. The following sections will focus on biomarker testing in oncology.
Biomarker testing (also known as mutation, genomic, or molecular testing) uses laboratory tests to help the health care team gather as much information as possible about a patient’s cancer and uncover whether the patient has an actionable mutation. Four categories of biomarker testing include: predisposition, diagnostic, prognostic, and predictive:

A companion diagnostic (CDx) is typically an in vitro diagnostic that detects a predictive biomarker to determine the likely efficacy and safety of a corresponding drug or biological product. For example, FoundationOne® CDx is an FDA-approved, tissue-based broad CDx that physicians can use to analyze the genomic profile of individual patient’s tumors. Results may help inform treatment decisions for over 20 FDA-approved targeted therapies. Clinicians should order the corresponding CDx to determine appropriate therapeutic options for each patient.
Biomarker testing can help providers make more informed treatment decisions based on a patient’s genomic makeup or the genomic makeup of their cancer. Additionally, biomarker testing can align with a health system’s broader strategic goals. For example, with the shift to value-based care, many organizations are focused on delivering appropriate, high-value care. Additionally, many organizations are focused on expanding their precision medicine programs to provide patients with more informed, personalized care. Biomarker testing aligns with each of these system-level strategic priorities.
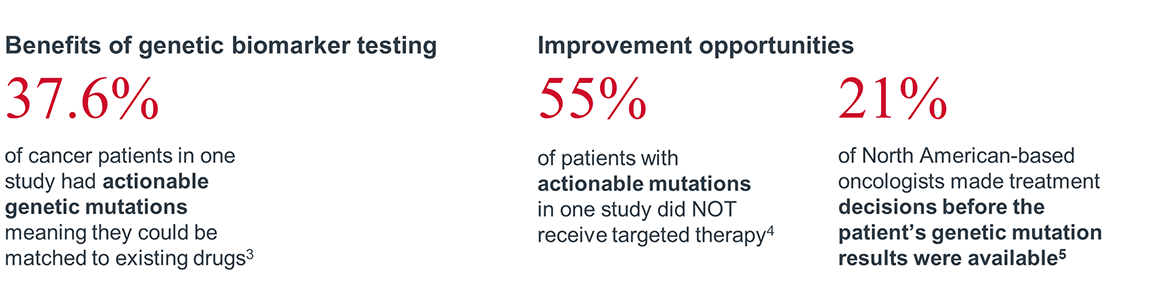
Given the advantages of biomarker testing, the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) recommend assessment of biomarker test results prior to treatment initiation in eligible patients with metastatic non-small cell lung cancer (mNSCLC). As a result, biomarker testing is essential to provide guideline-concordant care in many areas of oncology
Despite these benefits, gaps still exist in realizing the full potential of biomarker testing. Many patients who are eligible for biomarker testing do not receive guideline recommended testing. Of patients who do receive biomarker testing, many eligible patients do not receive targeted therapy due to challenges throughout the process (e.g., insufficient tissue, long turnaround times that prevent clinicians from using results to help inform treatment decisions, complicated or inaccessible test reports).
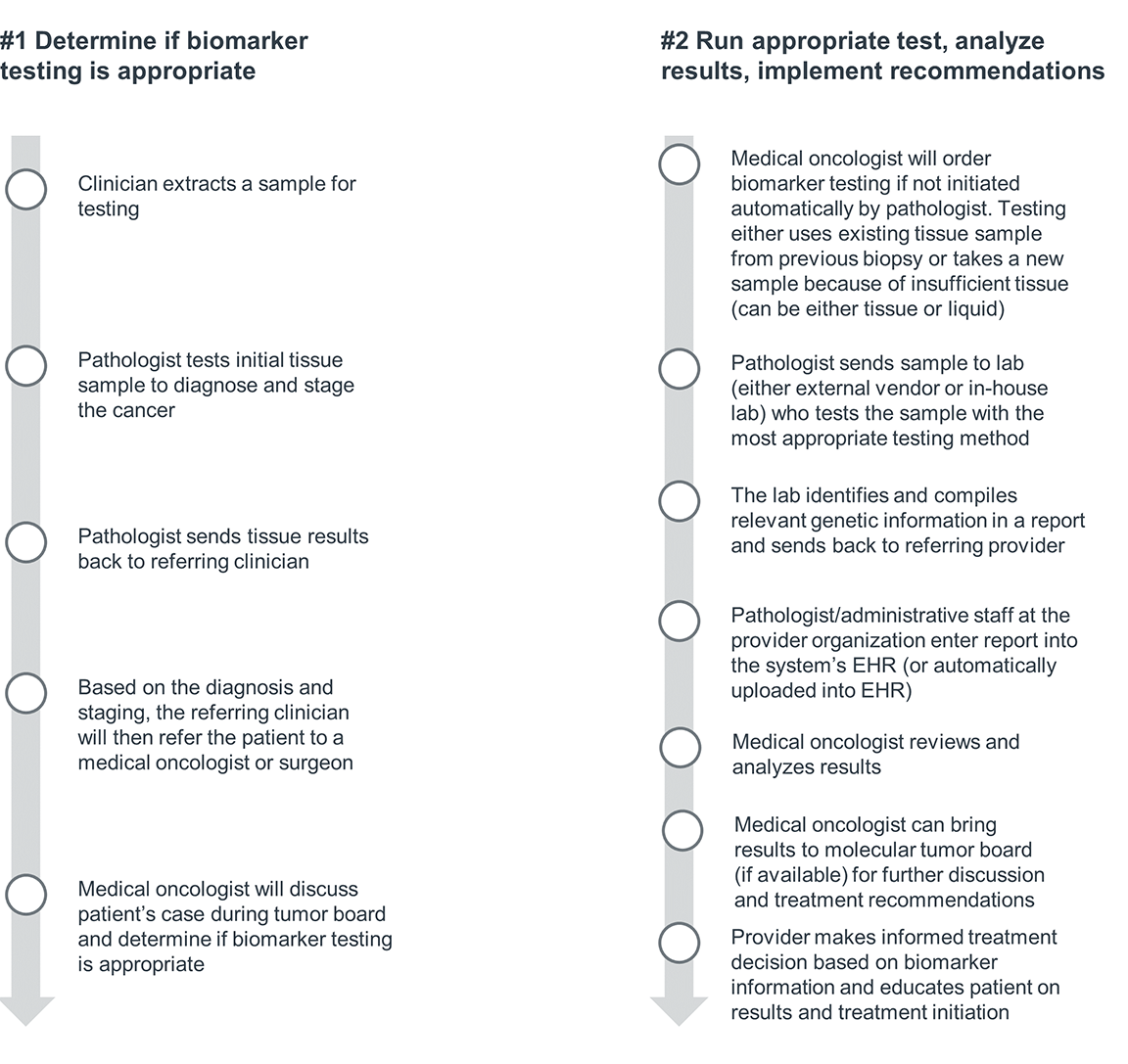
No two patient journeys look the same—especially in cancer care. However, the biomarker testing process does follow a general order of operations:
Conversations you should be having with your leadership team
Have you considered how biomarker testing can support our broader organizational goal of delivering high-value, appropriate care?
What are long-lasting, sustainable investments we can make in our biomarker testing process to support our push to precision medicine?
How can we standardize biomarker testing across our broader organization?
What internal resources (e.g., EHR, staff) can we leverage to improve our biomarker testing process?
Conversations you should be having with your frontline staff
What challenges do you run into when it comes to biomarker testing? What do you think works well?
How can we standardize our biomarker testing process across all our clinicians and sites?
How can we codify our processes and procedures to facilitate widespread adoption of those standards?
How can we broadly communicate the value of biomarker testing and provide updates on changing guidelines throughout our sites?
AstraZeneca focuses on the discovery, development and commercialisation of prescription medicines in Oncology and BioPharmaceuticals, including Cardiovascular, Renal & Metabolism, and Respiratory & Immunology.
This cheat sheet is sponsored by AstraZeneca for educational purposes only. The content, views, and opinions contained within the cheat sheet are copyrighted by Advisory Board and all rights are reserved. Advisory Board experts wrote the content, conducting the underlying research independently and objectively. Advisory Board does not endorse any company, organization, product or brand mentioned herein.

This report is sponsored by AstraZeneca. Advisory Board experts wrote the report, conducting the underlying research independently and objectively. AstraZeneca had the opportunity to review the report.
Learn moreDon't miss out on the latest Advisory Board insights
Create your free account to access 1 resource, including the latest research and webinars.
Want access without creating an account?
You have 1 free members-only resource remaining this month.
1 free members-only resources remaining
1 free members-only resources remaining
You've reached your limit of free insights
Become a member to access all of Advisory Board's resources, events, and experts
Never miss out on the latest innovative health care content tailored to you.
Benefits include:
You've reached your limit of free insights
Become a member to access all of Advisory Board's resources, events, and experts
Never miss out on the latest innovative health care content tailored to you.
Benefits include:
This content is available through your Curated Research partnership with Advisory Board. Click on ‘view this resource’ to read the full piece
Email ask@advisory.com to learn more
Click on ‘Become a Member’ to learn about the benefits of a Full-Access partnership with Advisory Board
Never miss out on the latest innovative health care content tailored to you.
Benefits Include:
This is for members only. Learn more.
Click on ‘Become a Member’ to learn about the benefits of a Full-Access partnership with Advisory Board
Never miss out on the latest innovative health care content tailored to you.
